Electron revolve around the nucleus in certain fixed circular orbits in which they can continue revolving without gaining or losing energy. Quantization means that a quantity cannot change gradually and continuously to have any arbitrary value but changes only abruptly and discontinuously to have certain definite or discrete value. This explains why the energy decreases as we move from outer to inner shell. Hence, the energy of the electron becomes less and less. As the electrons come more and more close to the nucleus, the attraction increases and more energy is released. Since its value was already zero, hence now it becomes negative. As the electron move towards the nucleus, it experiences a force of attraction by the nucleus. 
Hence, the energy of the electron at distance infinity from the nucleus is taken as zero. When the electron is at distance Infinity from the nucleus, there is no force of attraction on the electron by the nucleus. 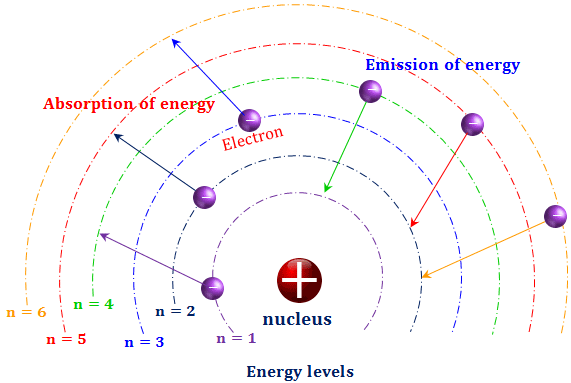
Where E 2 and E 1 are the energy of the electron in the higher and lower energy levels ΔE is the difference in energies of the two levels.Įnergy is always a emitted or absorbed in certain discrete quantities called quantas or photons and not any value. The amount of energy emitted or absorbed is given by the difference of energies of the two energy levels concerned i.e. This state of atom is called excited state .Since the lifetime of the electron in the excited state is short, it immediately jumps back to the lower energy level by emitting energy in the form of light of suitable frequency or wavelength. When energy is supplied to an atom ,an electron in the atom may jump from its normal energy level to some high energy level by absorbing a definite amount of energy. This state of atom is called normal or ground state.Ħ) Energy is emitted or absorbed only when the electron jumps from one orbit to the other. The angular momentum of an electron in an atom is also quantized.ĥ) When the electrons in an atom are in their lowest energy state, they keep on revolving in the respective orbits without losing energy because energy can neither be lost nor gained continuously. Where m is the mass of the electron, v is the tangential velocity of the revolving electron, r is the radius of the orbit, h is the Planck’s Constant and n is any Integer. The only permissible values of angular momentum are given by the expression: The energy of an electron is quantized.Ĥ)Like energy, the angular momentum of an electron in an atom can have certain definite or discrete values and not any value of its own. 
Where a 0= 5.29 pm is the radius of the first stationary state and is called Bohr’s radius.įor hydrogen like particles ,the radii of the stationary state are given by the expression:ģ) Since the electrons revolve only in those orbits which have fixed value of energy ,hence electrons in an atom can have only certain definite or discrete values of energy and not any value of their own. The energy of the levels increases as we move outwards starting from the first level.ī) The radii of the stationary states of the hydrogen atom are given by the expression: The first energy level(n=1) which is closest to the nucleus has lowest energy. Where Z is the atomic number and n stands for energy level. starting from the shells closest to the nucleus.Ī) The energies of the different stationary states in case of hydrogen atom are given by the expression The different energy levels are numbered as 1,2,3,4…. The energy of the electron revolving in a particular orbit is fixed and does not change with time. These orbits are called energy levels or stationary States. 1 Postulates of Bohr’s model of an Atomġ) An atom consists of a small, heavy positively charged nucleus in the centre and electrons revolve around it in circular orbit.Ģ) The electrons revolve only in those orbits which have a fixed value of energy.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
